

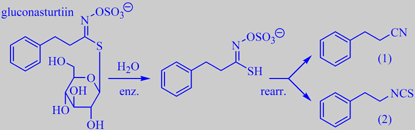
enzymatic cleavage of gluconasturtiin
Watercress
Nasturtium officinale (Cruciferae / Brassicaceae)
Watercress is a hardy perennial herb native to Europe and temperate Asia.
It loves slowly flowing clear brooks, and was cultured by the Romans in
irrigated gardens. Today it is mostly used as a culinary herb.
A characteristic of the Cruciferans is the so-called mustard oil glycosides,
in watercress represented by gluconasturtiin. By crushing (chewing) the
fresh leaves, gluconasturtiin is hydrolytically cleaved by the enzyme
myrosinase, and the reaction product spontaneously rearranged into hydrocinnamonitrile
(1) and
phenethyl isothiocyanate (2), the character compounds behind the pungent-cinnamic,
sharp-green aroma of watercress [162].
Etymology: Lat. Nasturtium, from Lat. nasus, nose; Lat.
torqueo, turning, torturing; Lat. officinale, useful
as a medicinal plant.


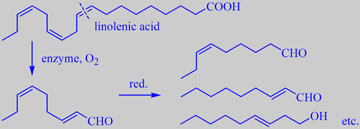
formation of C9 metabolites from linolenic acid
Citrullus lanatus (Cucurbitaceae)
Watermelon is a native of South Africa, but is now cultured all over the world in warm and dry climates.
Numerous sorts exist, and lately seedless varieties have been developed (photo). A chilled watermelon is a blessing on a hot summer day. The watery, sweetish flesh has aroma notes in common with cucumber, although less green and with a fruity shading.
Like in cucumber, the most important aroma compounds in watermelon are C9 metabolites of linolenic acid. Beaulieau and Lea found that (E,Z)-2,6-nonadienal, (E)-3-nonen-1-ol, (E)-2-nonenal, and (Z)-6-nonenal were the most abundant compounds in five varieties of seedless watermelons [148].
Fresh, watermelon-like notes have been popular in perfumery for several years.
Two aldehydes employed for this purpose should be mentioned:
Melonal, an extremely powerful and diffusive odorant with a green-citrusy, melon-like odour, and Methoxymelonal, equally diffusive, but milder and with a very beautiful, transparent watermelon character.
Melonal, for example, is used in L'Eau d'Issey (Miyake 1992) [43].
Melonal
2,6-dimethyl-5-heptenal
Methoxymelonal
2,6-dimethyl-6-methoxyheptanal


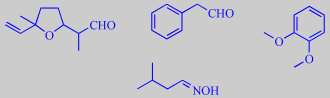
lilac aldehyde, phenylacetaldehyde, veratrole and isopentylaldoxime
White campion
Silene latifolia ssp. alba (Caryophyllaceae)
The flowers of white campion are closed in clear daylight but open in
the evening. They are then brightly white and fragrant during the night
with a characteristic scent. Pollinators are various moths. The Lychnis
moth (Hadena bicruris) takes part in a so-called nursery pollination
system with white campion. Its larvae feed on the seeds of the plant whereas
the adult moths perform the pollination.
Dötterl S et al. investigated in depth the role of specific odorants
of white campion flowers upon Lychnis behaviour and found that especially
lilac aldehyde, but also phenylacetaldehyde and veratrole were strong
attractors. Among 58 compounds identified, isopentylaldoxime or 3-methylbutanal
oxime was also characterized as a typical scent compound of white campion
(see also Honeysuckle
for floral aldoximes) [353].



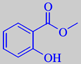
methyl salicylate
Wintergreen
Gaultheria procumbens (Ericaceae) Eastern Teaberry, Checkerberry
This tiny, North American plant of the heath family grows in pine forests
and on humid ground with sphagnum. The Mohicans and other Indians knew
its effects, which are due to gaultherin, a glucose-xyloside of methyl
salicylate [113]. Like salicylic acid, methyl salicylate is antiseptic,
analgesic and antipyretic. The whole plant smells of this compound, and
formerly the so-called Gaultheria oil or wintergreen oil was obtained
from it by steam distillation.
Etymology: Gaultheria, after the Canadian Dr. Gaultier, a botanist
from Quebec; Lat. procumbo, leaning forwards.
See meadowsweet
for more about salicylic acid.
Methyl salicylate has a fresh, sweet-phenolic odour. In Europe it is mostly known from sanitary products. In USA, on the contrary, methyl salicylate is used as a flavour compound in chewing gum and root beer.
The higher salicylates have quite another odour character. They are much appreciated perfumery ingredients. Isoamyl salicylate, for instance, has an intense, greenish, clover-like, balsamic odour. In combination with isoeugenol, it formed the basis of the successful perfume Trèfle Incarnat (Piver 1896). Today it is often replaced by
3(Z)-hexen-1-yl salicylate, having a more natural character. Another salicylic acid ester, benzyl salicylate, can be smelled from many flowers, e.g. carnation, Dianthus caryophyllus (Caryophyllaceae), and ylang-ylang, Cananga odorata (Annonaceae). It is used in perfumes of the L'Air du Temps type (Nina Ricci 1948).
See also Ashoka!
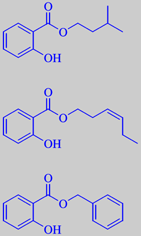
isoamyl salicylate,
3(Z)-hexenyl salicylate and
benzyl salicylate


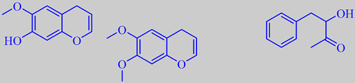
7-hydroxy-6-methoxy-4H-1-benzopyran, 6,7-dimethoxy-4H-1-benzopyran
and 3-hydroxy-4-phenyl-2-butanone
Wisteria sinensis (Fabaceae) Chinese wisteria
This climbing tree from South-East Asia is a common ornamental plant. In temperate gardens it thrives on a sheltered wall facing south. The fragrance of the wisteria flowers has fascinated perfumers for a long time - a pleasant, mild, warm and creamy sweetness with rosy, peppery and spicy nuances. Joulain et al. showed that the key ingredients of the characteristic fragrance are two unusual aromatic compounds, the beta-chromenes 7-hydroxy-6-methoxy-4H-1-benzopyran and 6,7-dimethoxy-4H-1-benzopyran [207]. Moreover, 3-hydroxy-4-phenyl-2-butanone or phenylacetoin has been shown to be present [114].